As of 2022, approximately 1.2 million people in the United States are living with HIV. Antiretroviral therapies have enabled many of these individuals to live productive, symptom-free lives, but a cure that permanently eliminates HIV from an infected person’s body is still a long way off. However, researchers at The Wistar Institute, an international biomedical research institute in the areas of cancer, immunology, infectious disease, and vaccine development, have zeroed in on a promising compound that targets HIV reservoirs that persist in people living with HIV despite the presence of anti-HIV therapy.
In a recent paper published in Antimicrobial Agents and Chemotherapy, researchers identified hopeaphenol, a natural plant-based compound, as possessing antiviral properties that are effective against HIV. Specifically, the compound helps to block not only viral replication but also to inhibit reactivation of the “viral reservoir” that persists after anti-HIV therapy within human immune cells and can make new virus at any time, even when patients are receiving ART and exhibit no viral symptoms.
“This is important because anti-HIV therapy can stop the symptoms, but it doesn’t eliminate the potential of the underlying HIV reservoir from re-emerging. The virus is still there and still a little bit active—kind of rumbling and turning on—and the immune system is stressed about that,” said Ian Tietjen, Ph.D., the lead author on the paper and a research assistant professor in the laboratory of Luis Montaner, D.V.M., D.Phil., senior author, in Wistar’s Vaccine & Immunotherapy Center.
Tietjen explained that the expression of persistent HIV can stress immune cells even in the presence of anti-HIV therapy, which has been linked to inflammation and higher risk of cancers, metabolic disease, heart disease, and other conditions associated with advanced aging in persons living with HIV.
“So, we think that something like hopeaphenol, which we’ve identified, could prevent the HIV reservoir from reactivating—reducing stress on the immune system and potentially decreasing these age-related conditions.”
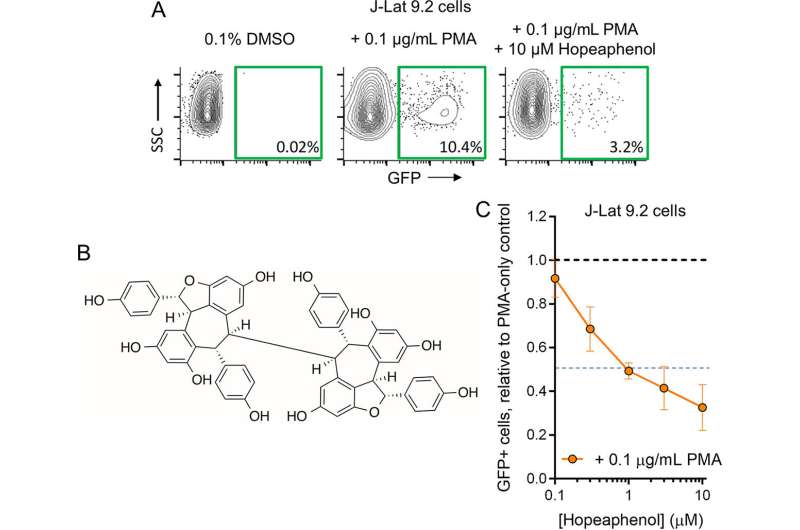
Tietjen discovered hopeaphenol’s potential for suppressing HIV expression by first testing a collection of 512 compounds. Hopeaphenol proved to be the most active, so they performed some additional testing. With the help of students from Cheyney University and Philadelphia area high school students participating in Wistar’s Education and Training Programs, they were able to verify their results and to analyze how the compound was working. Then, they conducted two experiments to demonstrate hopeaphenol’s therapeutic potential against HIV.
In the first experiment, Tietjen and his colleagues isolated lymphocytes (white blood cells) from human blood and infected those cells with HIV in the laboratory. They let the virus replicate and then treated the infected cells with hopeaphenol. After treatment, the virus stopped replicating.
In the second experiment, the researchers took CD4+ T cells—i.e., “helper T-cells”—from several people living with HIV taking anti-HIV therapy and divided the cells into two groups. One group of helper T-cells was pre-treated with hopeaphenol; the other group (the control) was left untreated. Then, Tietjen and his colleagues activated the cells’ that had viral reservoirs to start producing virus. They found that cells pre-treated with hopeaphenol reduced viral production—indicating that hopeaphenol was able to dampen viral reactivation.
“These observations suggest to us that hopeaphenol, in addition to just stopping active replication and spread, might also help silence the HIV reservoir by inhibiting viral reactivation not otherwise accomplished by current anti-HIV medicines,” said Tietjen.
Prior research has demonstrated that hopeaphenol is well tolerated in animal models, and because it can be derived from a variety of plants, its isolation can be scaled up easily. Due to these factors and his study’s results, Tietjen is optimistic that hopeaphenol can eventually contribute to a better quality of life for patients with HIV, although further study is still needed before it can be tested in humans.
“I think in time it could become a promising additive on top of existing antiretrovirals as a more potent anti-HIV therapy,” he said.
More information:
Ian Tietjen et al, The Natural Stilbenoid (–)-Hopeaphenol Inhibits HIV Transcription by Targeting Both PKC and NF-κB Signaling and Cyclin-Dependent Kinase 9, Antimicrobial Agents and Chemotherapy (2023). DOI: 10.1128/aac.01600-22
Journal information:
Antimicrobial Agents and Chemotherapy
Source: Read Full Article