Immunogenetics describes the investigation of the genetics underpinning the immune system and its responses. Its overall goal is to identify the specific genes responsible for encoding immunological cells and molecules.
DNA. Image Credit: vitstudio/Shutterstock.com
Linking genetics with immune dysregulation
Immunogenetics is highly pertinent to a plethora of immune-related disorders as it enables researchers and clinicians to identify the underlying causes of immune system dysregulation and formulate an adequate treatment strategy.
Typically, such genetic abnormalities result in either an inability to appropriately counteract pathogenic infections, or in a predisposition towards autoimmune disorders or cancer. Through such investigations, our understanding of the basic mechanisms involved in immunity as well as those underpinning immune tolerance, dysregulation and disease has been vastly refined and improved.
Genetic aberrations underpinning B and T Cell deficiency
During immune responses, the primary role of B lymphocytes (or B cells for short) is to create antibodies that can both detect and target pathogens. A number of steps are involved in the development and production of B cells.
Firstly, the pre-B cell receptor (BCR) complex must be activated, leading to the stimulation of various cell signaling pathways. As well as this, each B cell must undergo rearrangements of its immunoglobulin gene during its development in order to produce a unique antibody molecule.
This process is fundamental to the core function of B cells as it allows for them to create antibodies in response to any invading pathogen, which is also specific to that pathogen. Given this information, it is unsurprising that autosomal recessive mutations in the pre-BCR complex and in signaling chains IG-alpha and Ig-beta can cause B cell deficiency.
Similar mutations in proteins involved in breaking and rejoining DNA during immunoglobulin gene rearrangement; e.g., recombinase-activating genes 1 and 2 (RAG1/2), also result in B cell deficiency.
Similar to the manner in which B cells must successfully undergo immunoglobulin gene rearrangement, T cells are required to rearrange their T cell receptor gene in order to properly mature. As such, mutations that affect this process are linked to T cell dysregulation and immunodeficiency disorders.
Perhaps the most well-known of these disorders is severe combined immunodeficiency (SCID), which is known to be associated with mutations in the aforementioned RAG1/2; genes which are also required to produce the double-stranded breaks needed for T cell receptor gene rearrangement.
Patients suffering from SCID are required to avoid all possible points of contact with infectious diseases as their immune system’s inability to appropriately combat such infections typically leads to life-threatening consequences.
Human major histocompatibility complex mutations linked with multiple neurological disorders
The human major histocompatibility (MHC) complex, also referred to as the human leukocyte antigen (HLA) complex, is responsible for the coding of cell-surface proteins which are essential in modulating both innate and adaptive immune responses such as inflammation regulation and in the complement system.
Studies have found that genes found within the HLA family (found on the sixth chromosome) account for the greatest component of the genetic risk for a wide array of neurological diseases. Examples of these disorders include multiple sclerosis, Parkinson’s disease, schizophrenia, Alzheimer’s disease, amyotrophic lateral sclerosis and myasthenia gravis.
Gene therapy – the end goal of immunogenetics?
Gene therapy describes any clinical intervention that seeks to alter or manipulate the expression of a gene, typically with the intention of curing or preventing a disease. This can be achieved through the use of various techniques such as the insertion of genetically altered plasmid DNA , viral vectors or bacterial vectors.
The bacterial CRISPR (clustered regularly interspaced short palindromic repeats), first discovered by Francisco Mojica, has been shown to play an essential role in the prokaryotic adaptive immune system in conjunction with the Cas9 gene.
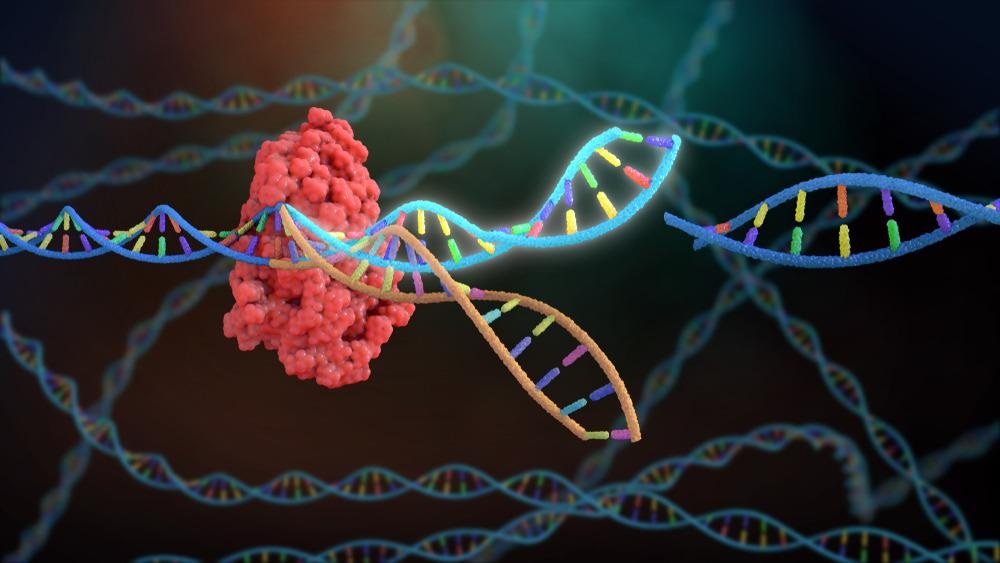
CRISPR. Image Credit: Nathan Devery/Shutterstock.com
The CRISPR sequences are derived from bacteriophages that have previously infected the prokaryote and are in turn used to combat further infections through the detection and destruction of similar bacteriophage DNA. CRISPR/Cas9 gene-editing takes this process and applies it to eukaryotic genomics.
It is a two-component system, the first being the single-effector Cas9 protein which is responsible for creating double-stranded breaks in the target DNA. The second component is a single guide RNA (sgRNA) which anchors to Cas9.
This sgRNA contains a 20 base pair spacer sequence which is specific to the target gene, allowing it to guide the CRISPR/Cas9 complex to the desired genomic location. To date, CRISPR has been successfully used to alter mosquito DNA to prevent their ability to transmit DNA.
Immunogenetics offers a clearly defined route through which gene therapies such as CRISPR can be effectively implemented. Through the identification of genes whose dysregulation is involved in immune system-related disorders, immunogenetics can act as a guide for gene therapy which could theoretically be used to ameliorate symptoms or potentially cure certain diseases.
In the past number of years, gene therapy has been employed in the treatment of B both cell lymphoblastic leukemia and B cell lymphoma, as well as in various other non-immune system-related disorders. With more immunogenetic research, it is extremely likely that further gene therapy targets will be identified.
References
- Allenspach, E.J., Torgerson, T.R. (2013). Immunogenetics. Brenner’s Encyclopedia of Genetics, 2, 25-28. https://doi.org/10.1016/B978-0-12-374984-0.00770-1.
- Liang, G., & Bushman, F. D. (2021). The human virome: assembly, composition and host interactions. Nature Reviews Microbiology, 1-14. https://doi.org/10.1038/s41579-021-00536-5
- Maude, S. L., Laetsch, T. W., Buechner, J., Rives, S., Boyer, M., Bittencourt, H., … & Grupp, S. A. (2018). Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. New England Journal of Medicine, 378(5), 439-448. https://www.nejm.org/doi/full/10.1056/NEJMoa1709866
- Misra, M. K., Damotte, V., & Hollenbach, J. A. (2018). The immunogenetics of neurological disease. Immunology, 153(4), 399-414. https://doi.org/10.1111/imm.12869
- Raghavendra, P., Pullaiah, T. (2018). Chapter 7 – Pathogen Identification Using Novel Sequencing Methods, Advances in Cell and Molecular Diagnostics, Academic Press, 161-202. https://doi.org/10.1016/B978-0-12-813679-9.00007-5.
- Shkoporov, A. N., Clooney, A. G., Sutton, T. D., Ryan, F. J., Daly, K. M., Nolan, J. A., … & Hill, C. (2019). The human gut virome is highly diverse, stable, and individual specific. Cell host & microbe, 26(4), 527-541. https://doi.org/10.1016/j.chom.2019.09.009
Further Reading
- All Immunology Content
- What is Immunology?
- What does it mean to be Immunocompromised?
- Classical Immunology
- Clinical Immunology
Last Updated: Feb 21, 2022
.jpg)
Written by
Chris Traynor
Chris graduated from University College Dublin in 2020 with a Bachelor of Science degree, majoring in Neuroscience. Over the course of his undergraduate studies, he investigated various areas such as nervous system development, sensory neuroscience, and neurodegenerative disorders.
Source: Read Full Article