Evolving variants of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) have become a growing concern worldwide for their rapid transmission and ability to cause severe infection. New research led by Ronit Rosenfeld from the Israel Institute for Biological Research confirms evidence that specific monoclonal antibodies can bind to the receptor-binding domain of the B.1.1.7 and B.1.351 variants and effectively neutralize them.
The study “The neutralization potency of anti-SARS-CoV-2 therapeutic human monoclonal antibodies is retained against novel viral variants” is available as a preprint on the bioRxiv* server, while the article undergoes peer review.
Study design
The research team previously reported that certain monoclonal antibodies specific to the receptor binding and N-terminal domain such as MD65 successfully neutralized SARS-CoV-2 in vitro and in vivo studies.
The current study builds on those findings by measuring the binding ability of four SARS-CoV-2 neutralizing monoclonal antibodies —MD65, MD62, MD29, and BL6 — to four distinct epitopes of the spike receptor-binding domains. The receptor-binding domains contained six mutations commonly associated with variants of concern, including N501Y, S477N, P.1, and E484K.
They also evaluated the neutralizing potency of two monoclonal antibodies specific to the N-terminal domain of B.1.1.7 and the B.1351 variant. A biolayer interferometry analysis was used to assess binding aptitude.
Binding profile amongst monoclonal antibodies
The researchers found about 5-22% of binding lost due to five or six substitutions in the receptor-binding domain of SARS-CoV-2.
A significant binding reduction of 74% was observed with the K417N mutation by the MD62 monoclonal antibody. There was also a 17% binding reduction when paired with the MD65 antibody.
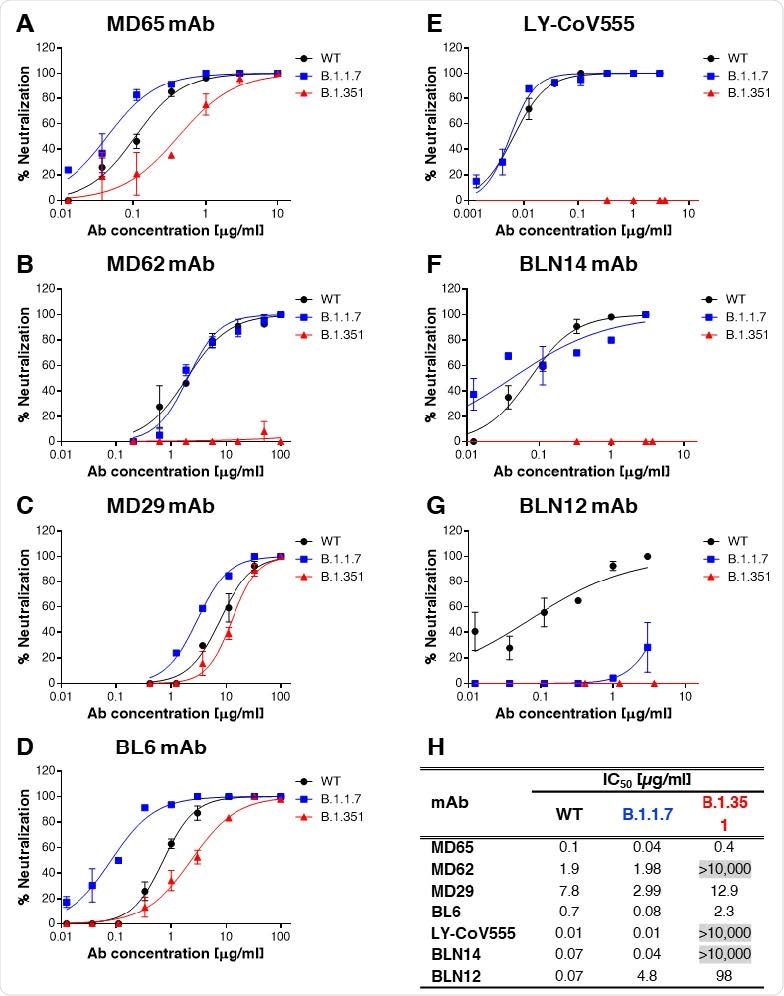
Of the 4 monoclonal antibodies specific to the receptor-binding domain, MD65 was the most effective in both neutralizing and providing long-term immunity at low doses.
Comparing epitope recognition with MD65 competitor
The LY-CoV555 monoclonal antibody potentially competes with the MD65 monoclonal antibody by binding to hACE2. Although the researchers note the two monoclonal antibodies do not have similar sequences, and their recognition patterns may differ despite targeting close epitopes.
To test this theory, the researchers evaluated LY-CoV555 binding against the SARS-CoV-2 spike protein and how it compared to the binding profile of MD65.
In the presence of the MD65 monoclonal antibody, the LY-CoV555 antibody could not bind to the rRBD protein suggesting both antibodies share epitope targets.
Against SARS-CoV-2 mutations, LY-CoV555 successfully recognized and bound to N439K, Y453Y, S477N, and N501Y.
However, the presence of the E484K substitution blocked LY-CoV555 binding to epitopes. “This observation is in line with recently reported studies, suggesting that the E484K substitution is accountable for the abolishment of neutralization of SARS-CoV-2 natural variants, carrying this mutation, by LY-CoV555 mAb,” wrote the researchers.
Binding ability against multiple SARS-CoV-2 mutations
The researchers used recombinant mutated spike S1 subunit proteins that were a mix of mutations found with the B.1.1.7 and B.1.351 variants. It contains the N501Y, K417N, and E484K mutation.
Monoclonal antibodies MD65, MD29, BL6, and LY-CoV555, were successful in binding to the B.1.1.7 recombinant spike protein. However, MD62 binding ability fell by about 45%. This suggests structural changes in the B.1.1.7 spike protein may have affected MD62 binding.
Binding reduction of LY-CoV555 and an 18% binding decrease in BL6 in the B.1.351 recombinant spike protein was likely due to the E484K substitution. The MD62 monoclonal antibody was unable to recognize and bind to the epitope, and there was a 65% binding reduction from MD65. The researchers suggest the decrease in binding ability is likely from the presence of the K417N substitution.
Immune escape potential from variants of concern
The researchers evaluated the ability of six monoclonal antibodies and LY-CoV555 to neutralize variants of concern that have been associated with escaping the immune system. Specifically, they evaluated their towards the N-terminal domain for B.1.1.7 and B.1.351 live variants.
The MD65, MD62, MD29, BL6, and LY-CoV555 were the most successful in neutralizing the B.1.1.7 variant. The researchers observed that MD65, MD29, and BL6 showed superior neutralization.
The B.1.351 variant showed a more robust immune escape potential than the B.1.1.7 variant. Similar to what was observed with the B.1.351 recombinant spike protein, MD62 and LY-CoV555 showed a complete loss of binding.
Fortunately, MD65, MD29, and BL6 effectively neutralized the B.1.351 variant.
Important Notice
*bioRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
- Makdasi et al. The neutralization potency of anti-SARS-CoV-2 therapeutic human monoclonal antibodies is retained against novel viral variants. bioRxiv, 2021. doi: https://doi.org/10.1101/2021.04.01.438035, https://www.biorxiv.org/content/10.1101/2021.04.01.438035v2
Posted in: Medical Research News | Disease/Infection News
Tags: Antibodies, Antibody, Coronavirus, Coronavirus Disease COVID-19, Genetic, Immune System, in vitro, in vivo, Monoclonal Antibody, Mutation, Protein, Receptor, Research, Respiratory, SARS, SARS-CoV-2, Severe Acute Respiratory, Severe Acute Respiratory Syndrome, Spike Protein, Syndrome

Written by
Jocelyn Solis-Moreira
Jocelyn Solis-Moreira graduated with a Bachelor's in Integrative Neuroscience, where she then pursued graduate research looking at the long-term effects of adolescent binge drinking on the brain's neurochemistry in adulthood.
Source: Read Full Article